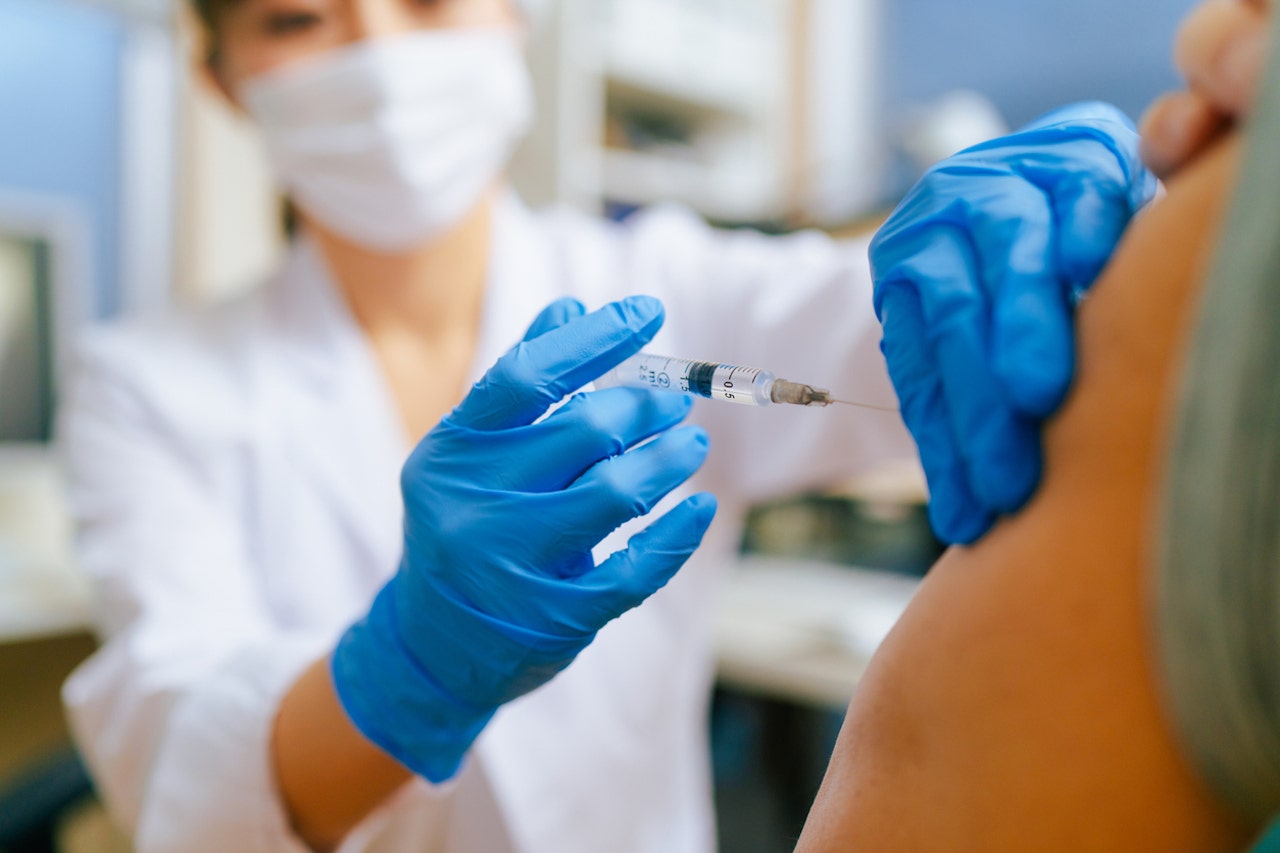
The US Food and Drug Administration (FDA) has updated Covid-19 vaccinations from Moderna and Pfizer for the 2024-2025 season.
The updated mRNA vaccines have been “authorized and authorized for emergency use,” according to an FDA statement released today.
The monovalent (single) vaccines target the omicron variant KP.2 of the SARS-CoV-2 strain.
RISKS RELATED TO THE COVID VACCINE IDENTIFIED IN THE STUDY
“These vaccines have been updated to provide better Protection against COVID-19 caused by circulating variants,” the FDA said.
“Vaccination remains the cornerstone COVID-19 prevention,” said Peter Marks, MD, Ph.D., director of FDA’s Center for Biologics Evaluation and Research, in the FDA announcement.

The U.S. Food and Drug Administration has approved updated COVID-19 vaccines from Moderna and Pfizer for the 2024-2025 season. (REUTERS/Andrew Kelly/File photo)
“These updated vaccines meet the agency’s rigorous scientific standards for safety, effectiveness and manufacturing quality.”
CLICK HERE TO SIGN UP FOR OUR HEALTH NEWSLETTER
“Given the declining immunity of the population from previous contact with the virus and from previous vaccinations, we strongly recommend that those eligible receive an updated COVID-19 vaccine to better protect against the currently circulating variants.”
The updated mRNA vaccines have been “authorized and approved for emergency use,” the FDA said in a statement. (iStock)
The CDC recommends that everyone ages 6 months and older receive the current COVID-19 vaccine.
CLICK HERE TO GET THE FOX NEWS APP
This also includes women who pregnant or breastfeeding.
During the week ending August 10, the Centers for Disease Control and Prevention (CDC) reported that 18.1% of COVID tests were positive.
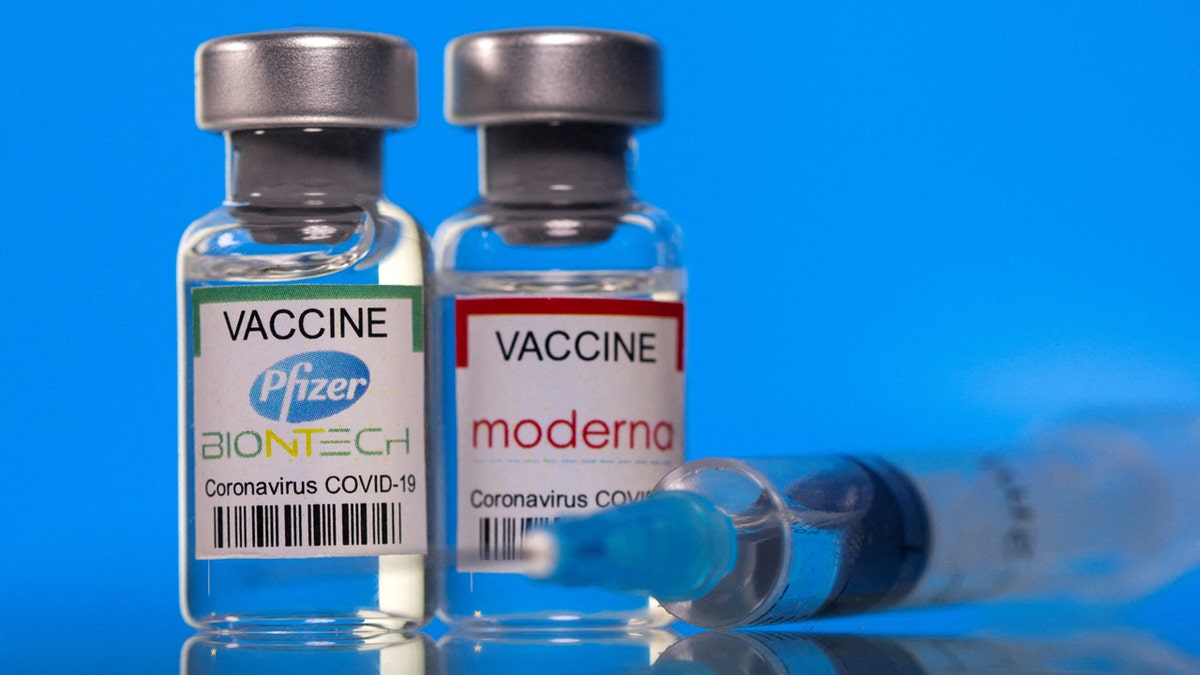
The monovalent (single) vaccines target the omicron variant KP.2 of the SARS-CoV-2 strain. (Reuters/Dado Ruvic/Illustration/File photo)
Meanwhile, 2.4% of patients in the emergency room were diagnosed with COVID-19 – a decrease of 1.5% from the previous week.
The proportion of COVID-related deaths was 1.9%, up from 1.6% the previous week, according to the CDC.
For more articles on health, see www.foxnews/health
Individuals should talk to your doctor if they have questions about the COVID-19 vaccine, the agency said.